Omicron iterates fast, can COVID-19 vaccine market learn from the "flu model"
BA.4 and BA.5, the new branches of Omicron with stronger communication power, have recently attracted attention. The new branch has triggered a new infection peak in South Africa, and the Portuguese National Institute of Health also estimates that its growth advantage is 13% more than that of BA.2.
Screenshots from the nextstrain website show that the different Omicron branches represented by yellow, orange and red are iterating rapidly within half a year, and the new branches represented by orange and red are also expanding in the cracks.
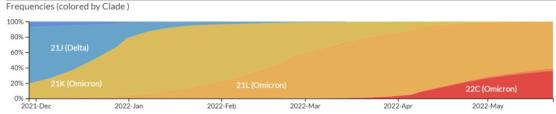
Screenshot of nextstrain website. According to the uploaded sequencing data, since December last year, the mainstream branches in Omicron have gradually changed from BA.1 (shown as 21K) to BA.2 (shown as 21L) to BA2.12.1 (shown as 22C), and the area occupied by new branches is also expanding.
The new branches of Omicron are frequent. Is the current COVID-19 vaccine still effective? Do the branches BA.2 and BA.5 need their own vaccines? Should the update of COVID-19 vaccine keep up with the variation of virus? How to keep up with the virus mutation? With the above questions, the reporter of Science and Technology Daily contacted and interviewed a number of industry experts.
Is the current vaccine still effective?
Does the mutation branch in Omicron constantly trigger new infection peaks, which means that the current COVID-19 vaccine will be invalid?
Authoritative experts in the industry responded to the Science and Technology Daily reporter and said: "Current vaccines can still provide the most basic protection in ensuring the reduction of severe illness and death.. The epidemic in COVID-19 has been going on for two and a half years. At present, it seems that there is no possibility for Covid-19 to achieve zero infection through vaccination. COVID-19 vaccine can ensure the effect of reducing infection and avoiding serious illness and death. "
Regarding the role of the current vaccine in epidemic prevention and control, the person in charge of vaccine-related production enterprises in COVID-19 holds the same view: "The existing vaccine is no problem in preventing severe illness and death at present, but it will not be 100% anti-infection."
Many statistics or research results from the real world show that the current vaccine plays a significant role in preventing severe illness and death. For example, the latest data in Hong Kong (as of May 25th) showed that the mortality rate of those infected in Omicron who were not vaccinated was 3.04%, while the mortality rate of those infected with three doses of vaccine dropped significantly to 0.04%.
Do the branches BA.2 and BA.5 all need their own vaccines?
According to WHO’s latest weekly report on epidemiological progress in COVID-19 (May 25th),Omicron BA.2 and its progeny pedigree (BA.2.X) have become the main popular variants in the world.At present, the relative proportions of BA.2.X, BA.4 and BA.5 are 94%, 0.8% and 1% respectively.
This shows that during the evolution of Omicron mutant, the branch BA.1 is disappearing. However, the reporter learned that at present, most vaccines against Omicron in China are designed and developed for BA.1.
Whether it is necessary to develop new vaccines for new branches, the industry generally judges from the perspective of vaccine protection: if the activity of neutralizing antibodies produced by the original vaccines against mutant strains is greatly reduced, it is necessary to develop new vaccines; If it still has a good neutralizing effect on the mutant, there is no need to develop a new vaccine.
Although Omicron strain mutates rapidly and different branches appear, butNew vaccines will not be introduced for each branch, and there is no need to do so.. Relevant experts reminded: "If you continue to monitor, you will find that many mutations will disappear soon."
Can vaccine update keep up with virus iteration?
In the technical route of vaccine development in COVID-19, the design cycle of mRNA vaccine is short. On the basis of a mature platform, the design can be completed within 7 days, and the preclinical trials including animal experiments can be completed in about one month.
Industry experts engaged in the research and development of mRNA vaccine said that for the variant branch, the mRNA vaccine does not need to obtain the strain, but only needs to optimize the vaccine sequence according to the variant sequence and test the expression efficiency, and then pre-clinical trials can be carried out. Covid-19 Omicron mutant mRNA vaccine, developed by Suzhou Aibo Bio, China, has been approved for clinical research in UAE.
In response to virus variation, the technical route of inactivated vaccine can also complete the preclinical study of new strains in a short time by establishing an emergency system. The reporter previously learned that it can be advanced to the clinical trial application stage about 2 months after obtaining the new strain.
Keep up with the variation, can COVID-19’s vaccine market learn from the "flu model"
In order to cope with the frequent mutation of viruses, there are precedents for vaccine updates to follow closely. For example, the influenza virus vaccine is recommended by WHO every year, and countries produce new influenza vaccines accordingly.
In order to keep up with the variation in Covid-19, can the "flu model" be used for reference in the marketing of vaccines in COVID-19?
“It is estimated that the future development of COVID-19 vaccine needs to adopt the influenza model.The authoritative experts mentioned above responded, but WHO is unable to recommend mainstream strains through monitoring now, because the pandemic is not over yet.
Another advantage of the "flu model" is that it greatly shortens the time to market for new vaccines. Because there is no need for re-approval, its batch issuance system can effectively shorten the period from vaccine development to marketing. Experts in the industry said that after polishing and construction, various technical routes and platforms have gradually matured, the research and development speed has been accelerated, and the production process has been stable.If the "flu model" can be adopted in the development and marketing of vaccines in COVID-19, the time to market for new vaccines will be greatly shortened.
WHO has revealed some unexpected phenomena in its weekly epidemiological report: available data show that BA.4, BA.5 and BA.2.12.1 spread faster in countries where BA.1 was once prevalent, but less in countries where BA.2 was prevalent; The vaccination degree of each country may have an impact on the popularity of the new branch of Omicron. Visible,Covid-19 mutation and vaccine immunogenicity are mutually causal and widely related, and updating the vaccine in time may have an impact on virus mutation.
Source: Science and Technology Daily
Original title: "Omicron iterates fast, is it necessary to be" anxious "about the vaccine? 》
Read the original text